Associations Between Indoor Air Pollutants and Risk Factors for Acute Respiratory Infection Symptoms in Children Under 5: An Analysis of Data From the Indonesia Demographic Health Survey
Article information
Abstract
Objectives
The study investigated the association between indoor air pollution (IAP) and risk factors for acute respiratory infection (ARI) symptoms in children under 5 years of age.
Methods
A cross-sectional study was conducted using data derived from Indonesia Demographic and Health Survey in 2017. Binary logistic regression modeling was employed to examine each predictor variable associated with ARI among children under 5 years of age in Indonesia.
Results
The study included a total of 4936 households with children. Among children under 5 years old, 7.2% reported ARI symptoms. The presence of ARI symptoms was significantly associated with the type of residence, wealth index, and father’s smoking frequency, which were considered the sample’s socio-demographic characteristics. In the final model, living in rural areas, having a high wealth index, the father’s smoking frequency, and a low education level were all linked to ARI symptoms.
Conclusions
The results revealed that households in rural areas had a substantially higher level of reported ARI symptoms among children under 5 years old. Furthermore, the father’s smoking frequency and low education level were associated with ARI symptoms.
INTRODUCTION
Various agents, such as viruses and bacteria, can cause acute respiratory infections (ARIs). Symptoms typically appear within a few hours to a few days after infection. ARI symptoms can include fever, body pains, runny nose, and cough [1]. The most common symptoms are throat irritation, coryza, shortness of breath, wheezing, or breathing difficulties. Pneumococcal bacteria and tuberculosis contribute to this disease, along with influenza and parainfluenza viruses, rhinovirus, respiratory syncytial virus, and severe acute respiratory syndrome coronavirus [1]. Among the Association of Southeast Asian Nations countries, Indonesia has the highest mortality rate for children under 5 due to ARIs. According to the Indonesian death survey, ARIs account for 22.30% of all under-5 mortality [2]. The 2018 Basic Health Research results indicate that toddlers represent the highest number of ARI cases [2]. In developing countries, children under 5 remain at risk of severe illness and death due to ARIs [3].
The World Health Organization estimates that about 3 billion people use solid fuels for heating and cooking in their homes. They cook and heat their homes using basic stoves and open flames, burning coal and biomass (plant waste, wood, and animal waste). Solid fuel cooking contaminates the air, leading to premature deaths for more than 4 million individuals due to related diseases [4]. Research indicates that particulate matter (soot) inhaled from household air pollution is responsible for over 50% of early mortality in children under 5 due to respiratory infections [5]. Furthermore, a significant body of research has demonstrated that exposure to biomass smoke from polluting fuels increases the incidence of acute respiratory illnesses in children under 5 in developing countries [2,3].
In addition to cooking fuel, smoking by mothers and other household members also influences the frequency of ARI symptoms in children [6.7]. Evidence from a study conducted in Ethiopia suggests that children from low-income families are more susceptible to ARIs [8]. According to other cross-country studies carried out in developing countries, working mothers, older children, and boys born underweight all had a higher chance of experiencing ARIs [9]. Several studies have shown that mothers’ poor economic status and low maternal education are significant risk factors for child ARI [8-12]. Moreover, research has also identified rural residence [13], water facilities, and sanitation [14] as additional risk factors for ARI symptoms in children. This study aims to determine the association between indoor air pollution (IAP) and the likelihood of ARI symptoms in children under the age of 5 in Indonesia.
METHODS
This cross-sectional study utilized data from the 2017 Indonesia Demographic and Health Survey (IDHS). Families with a child under 5 who had completed the Indonesian government’s basic vaccination program were included in the study. Children with incomplete family identity data and those born prematurely were excluded. The sample size consisted of 4953 households with children under 5 from the 2017 IDHS. All participants included in the IDHS were recruited following guidelines established by the Indonesian National Statistical Office.
IAP exposure was measured using solid fuel usage reporting (SFUR). The SFUR categorizes fuel materials into two groups: non-solid fuels (such as electricity, liquefied petroleum gas, natural gas, and biogas) and solid fuels (such as kerosene, coal, lignite, charcoal, wood, animal dung, straw/bush/grass) [15]. The father’s smoking status was also considered an indicator of IAP exposure and was categorized into 3 groups: daily smoking (code 1), occasional smoking (code 2), and non-smoking (code 3).
ARI variables were assessed by asking each mother if her child had experienced a cough within the 2 weeks prior to the survey. If the response was affirmative, the mother was asked if the cough had worsened due to shortness of breath or rapid breathing problems. The ARI outcome variable referred to children who had a cough accompanied by respiratory difficulties. The ARI variable was coded as 1 if both responses were “yes” (e.g., experiencing cough and respiratory difficulties) and 0 if both answers were “no” (e.g., no cough and no respiratory difficulties) [6,10].
The statistical analysis was conducted using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). A descriptive analysis was employed to summarize the main characteristics of the study participants. The chi-square test was used to display cross-tabulations, while logistic regression was utilized to evaluate relationships among the study participants. This study applied weighting for all data sets, with self-weighting samples having a variable equal to 1 000 000. The weighting method involved dividing the variable weight by 1 000 000 for individual female samples.
Control variables adjusted for in the analysis included residence types (urban, rural), number of people in the household (1-3 or ≥3), wealth index (poorest or poor, middle, or rich), type of toilet (flush or poor flush toilet, pit latrine, composting toilet, or other), types of drinking water (unimproved source or improved source), mother’s age (15-24, 25-34, or more than 35 years old), mother’s education level (no education, primary, secondary, or higher), number of pregnancies (1, 2, and 3), father’s age (15-24, 25-34, or more than 35 years old), father’s education level (no education, primary, secondary, or higher), child’s sex (male or female), child’s age in years (1, 2, or 3), and exclusive breastfeeding (yes or no).
In the multivariate analysis, the relationship between each predictor variable and ARI was examined using logistic regression modeling. Significance was determined using a p-value <0.05 and 95% confidence intervals (CIs). Potential components for the multivariate analysis were included based on bivariate analysis with a p-value <0.2 [16]. The first step involved incorporating household characteristics in the multivariate analyses, followed by the addition of parental characteristics to the final model. The final model consisted of all participant attributes, including those of the children. According to the variance inflation factor (VIF) test, the final regression model indicated no multicollinearity among all included variables (VIF values<10) [17].
Ethics Statement
Informed consent for participation in the study was obtained from all participants. In accordance with university requirements and approval for the use of secondary data, an institutional review board (IRB) application was submitted to the institution and granted in 2022. Ethical approval was obtained from the IRB via Letter No. 012207091 dated July 20, 2022, and informed written consent was obtained from all parents in the study group.
RESULTS
A total of 4953 families with children under 5 were included in the study. In terms of weighting, the majority of households were located in rural areas (50.9%), with 1 to 5 members (65.1%) per home. Most households had a middle wealth index (79.7%), used non-solid fuel for cooking (76.8%), relied on unimproved drinking water sources (59.3%), and utilized flush or poor flush toilets (86.9%).
The results demonstrated that 60.5% of mothers and 60.0% of fathers had completed senior high school. Most parents were between 25 years old and 34 years old (52.6% for mothers and 46.9% for fathers). A total of 88.7% had a parity of 1-3 children, and 98.8% reported that the father in the household smoked cigarettes daily. Additionally, the proportion of girls (51.7%) was higher than that of boys (48.3%), and the majority of children were exclusively breastfed (87.9%). The findings also revealed a 7.2% prevalence of ARI symptoms among children under 5. The distribution of participants’ characteristics is presented in Table 1.
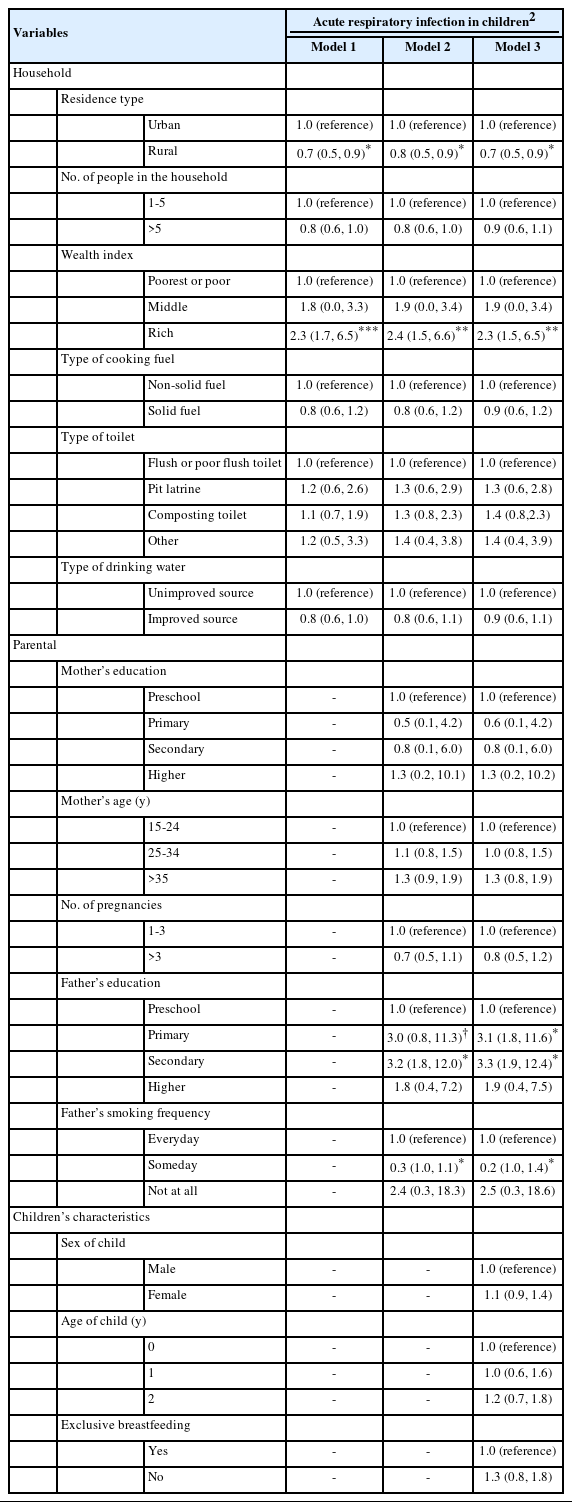
Table 2 displays the results of the bivariate analyses examining the relationship between various socio-demographic characteristics and ARI symptoms in children under 5. The associations were reported as p-values with 95% CIs. Among the socio-demographic factors, ARI symptoms were significantly related to the type of residence, wealth index, and father’s smoking frequency. Children living in rural areas and with fathers who smoked had a higher likelihood of reporting ARI symptoms. Moreover, households with a middle wealth index score were less likely to report their children experiencing ARI symptoms.
Table 3 summarizes the findings from the multivariate analyses. In model 1, children from rural families were more likely than those from urban families to report experiencing ARI symptoms. Additionally, households with a rich wealth index were associated with ARI symptoms. In the second and third models, a similar trend was observed. Families in rural areas reported ARI symptoms more frequently than those in urban areas. Families within the richest and rich wealth index scores had a higher risk of reporting that their children experienced ARI symptoms. Furthermore, smoking habits and having a loweducated father were linked to incidences of ARI symptoms in children under 5.
DISCUSSION
This is one of the most comprehensive surveys investigating the association between exposure to IAP and ARI symptoms in children under 5 using data from IDHS. We found that ARI symptoms were significantly correlated with residing in rural areas. This result was expected because people in rural areas tend to live in dry, dusty environments, whereas in cities, dusty environments may be limited to areas with industries and urbanization. According to reports, dry seasons have a greater impact in rural than urban regions, increasing the incidence of ARI symptoms, particularly in preschoolers [18,19]. A study conducted in Ethiopia also found ARI prevalence to be independently and significantly correlated with geographical areas [20].
We observed that households with medium income levels were associated with ARI symptoms among children under 5. Furthermore, in low-income households, there may not be a dedicated kitchen, so cooking may occur in open areas with natural ventilation, reducing exposure to IAP, even though solid fuels might be more common. In contrast, middle-income households may have a dedicated cooking space inside their homes, which increases exposure to IAP [21]. Poor households may have fewer domestic animals and livestock, which could increase the risk of infection, potentially explaining why medium-income families were more likely to report childhood ARIs than poor households [22]. In contrast to other studies, many families lived in substandard housing or remained exposed to toxins, pollutants, and infections due to low incomes and occupational constraints [23-25]. Previous research found that children who were in the kitchen while food was being cooked were twice as likely to develop symptoms of ARIs as those who were not in the kitchen [26].
There is evidence that exposing children to environmental cigarette smoke during pregnancy or postpartum can harm their immunological and respiratory systems, cognitive development, and lung function later in life [27-30]. We found a significant association between smoking frequency and ARI symptoms. Several studies have also found that paternal or familial smoking was a significant predictor of ARI symptoms [31,32]. In an earlier study, ARI symptoms varied considerably based on maternal smoking behavior. Children born to mothers who smoked regularly in the previous 24 hours exhibited significantly different ARI symptoms than children whose mothers did not smoke or smoked no more than 6 times [32].
If a family member smokes without being aware of their surroundings, it can have serious consequences for both the smoker and those around them, including toddlers with ARI symptoms [33]. Additionally, Tazinya et al. [34] found in a hospital in Cameroon that smokers had a higher risk of experiencing ARIs than non-smokers. Choube et al. [35] reported that toddlers whose families smoked indoors had a higher incidence of ARIs. Children are more sensitive to airborne pollutants (e.g., cigarette smoke) because their lungs are less capable of protecting against inhaled particles due to their higher breathing rate relative to their body size.
Education level was also identified as an important factor related to ARI symptoms among children under 5 years old. This study observed a significant association between the father’s education level and reported children’s ARI symptoms. Similarly, a survey from Gorkha, Nepal found a significant association between the father’s educational status and the prevalence of ARI in under-5 children [36]. Since an individual’s education is essential for making informed decisions about their health, the literacy level influences how individuals become aware of and access health information [37]. Parental education plays a vital role in ensuring children’s health quality. The present study showed no significant association between ARI symptoms in children and age, sex, type of toilet used, drinking water sources, exclusive breastfeeding, number of siblings, or marital status as reported by the households. However, several studies have revealed strong correlations between socio-demographic characteristics and symptoms [19,26,38].
Similar research has been carried out by Windi et al. [39]. They used all IDHS data and included 49 627 females and 15 993 children under the age of 5. According to that study, socio-demographic factors are important drivers of ARI among children under 5 in Indonesia. The authors found a strong correlation between ARI and a child’s age, mother’s profession, wealth index, and region of residence in Indonesia. The research demonstrated the significance of the wealth index of families and neighborhoods in improving children’s health outcomes in Indonesia [39]. In addition, another study used data from the Sleman Health and Demographic Surveillance System, including 463 children, to determine the factors associated with ARI in children under 5 in Sleman, Yogyakarta, Indonesia. They found that the risk of ARIs increased among respondents who used tap water or refilled water depots [40].
This study has several strengths. First, the IDHS is nationally representative and has a high response rate (97-99%) [16]. Second, a standardized questionnaire was used, which has been validated by many previous research studies [16]. However, there are also some limitations. First, since this study used existing datasets, it was not possible to include some potentially confounding variables. Second, the collection of self-reported information by mothers is inevitably prone to recall bias. However, this study selected data from families with a child under 5 who had completed the Indonesian government’s basic vaccination program, with 4953 households with children under 5 included. Additionally, this study did not collect information on specific ventilation configurations; therefore, we did not analyze the association between ventilation and ARI symptoms.
In conclusion, this study revealed that households in rural areas were significantly associated with a higher occurrence of ARI symptoms in children under 5. Additionally, the frequency of the father’s smoking was linked to ARI symptoms.
Notes
CONFLICT OF INTEREST
The authors have no conflicts of interest associated with the material presented in this paper.
FUNDING
This project was supported by Research Institute and the Community Service, Universitas Ahmad Dahlan, Yogyakarta, Indonesia (No. PDP-081/SP3/LPPM-UAD/VII/2022).
AUTHOR CONTRIBUTIONS
Conceptualization: Nurullita Santri I, Wardani Y. Data curation: Nurullita Santri I, YW, Vincent Abero Phiri Y, Suryani D. Formal analysis: Nurullita Santri I. Funding acquisition: Nurullita Santri I. Methodology: Nurullita Santri I. Writing – original draft: Nurullita Santri I. Writing – review & editing: Nurullita Santri I, Wardani Y, Vincent Abero Phiri Y, Nyam G, Aisyah Putri T, Isni K, Suryani D, Sambo G.
ACKNOWLEDGEMENTS
We acknowledge Indonesia Demographic and Health Survey (IDHS) for granting us access to the data set used in this study.