Association of Selected Medical Conditions With Breast Cancer Risk in Korea
Article information
Abstract
Objectives
To estimate the effect of medical conditions in the population of Korea on breast cancer risk in a case-control study.
Methods
The cases were 3242 women with incident, histologically confirmed breast cancer in two major hospitals interviewed between 2001 and 2007. The controls were 1818 women each admitted to either of those two hospitals for a variety of non-neoplastic conditions. Information on each disease was obtained from a standardized questionnaire by trained personnel. Odds ratios (ORs) for each disease were derived from multiple logistic regression adjusted for age, age of menarche, pregnancy, age of first pregnancy, and family history of breast cancer.
Results
Among all of the incident breast cancer patients, pre-existing diabetes (OR, 1.33; 95% confidence interval [CI], 0.99 to 1.78), hypertension (OR, 1.46; 95% CI, 1.18 to 1.83), thyroid diseases (OR, 1.26; 95% CI, 1.00 to 1.58), and ovarian diseases (OR, 1.70; 95% CI, 1.23 to 2.35) were associated with an increased risk of breast cancer when other factors were adjusted for. In a stratified analysis by menopausal status, pre-existing hypertension (pre-menopause OR, 0.80; 95% CI, 0.48 to 1.34 vs. post-menopause OR, 1.87; 95% CI, 1.44 to 2.43; p-heterogeneity <0.01) and ovarian disease (pre-menopause OR, 4.20; 95% CI, 1.91 to 9.24 vs. post-menopause OR, 1.39; 95% CI, 1.02 to 1.91; p-heterogeneity 0.01) showed significantly different risks of breast cancer.
Conclusions
Our results suggest the possibility that medical conditions such as hypertension affect breast cancer development, and that this can differ by menopausal status. Our study also indicates a possible correlation between ovarian diseases and breast cancer risk.
INTRODUCTION
Breast cancer is the second most common cancer among Korean women [1]. Multiple factors are associated with an increased risk of breast cancer, including age [2], gender [3], ethnicity [4], past history of breast cancer [5], reproductive and hormonal factors [6], family history and genetic factors [7], exposure to ionizing radiation [8], and environmental and lifestyle factors [9,10]. A high estrogen level is suspected to increase breast cancer risk [11], and other hormones such as progesterone, androgen derived from the ovaries and adrenal, thyroid hormones and insulin may play important roles in breast cancer development [11,12].
Certain medical conditions, such as hypothyroidism, diabetes, metabolic syndrome, obesity, hyperlipidemia, endometriosis, and ovarian cysts, have been reported to increase the risk of breast cancer associated with these altered hormone levels [13-16]. In addition, some diseases with an inflammatory component by viral or bacterial infection or an auto-inflammatory sequence are known to frequently progress to cancer [17]. Menopause brings great changes to a woman's endocrine system, and the carcinogenesis process may differ in women with the same medical conditions depending on their menopausal status. However, evidence for the associations between previous diseases and breast cancer development according to menopausal status is lacking.
Although some of these studies illuminate the relationship between certain medical conditions and the risk of breast cancer, whether most pre-existing diseases increase or decrease the risk of breast cancer in the Korean population has not been fully or precisely addressed. In this study, pre-existing medical conditions showing a relatively high prevalence in the Korean population were selected according to their hormonal or inflammatory characteristics. The present analysis used data from a hospital-based case-control study to estimate the effect of medical conditions prevalent in a Korean population on breast cancer risk.
METHODS
The parent study was the Seoul Breast Cancer Study (SeBCS), which is a multicenter case-control study of incident breast cancer patients and controls recruited from hospitals or the community that has been conducted beginning in 1995. The design and the characteristics of the participants have been previously described in detail [18,19]. Because of the lack of availability of pre-existing information on some participants, only 3486 cases and 1894 controls were used in this study. After excluding participants with a previous cancer history from the interview in both the cases and controls, 3242 cases and 1818 controls were used for the final analysis.
The participants were interviewed with a standardized questionnaire by trained interviewers for items such as demographic characteristics, menstrual and reproductive history, family history of breast and other cancer, body size, physical activity, alcohol consumption, smoking habit, and information on pre-existing medical conditions. The status of menstruation was determined by asking the following question: "Do you have regular menstruation now or have you experienced menopause?" To determine pre-existing medical conditions, 8 items were asked: whether participants had had tuberculosis, diabetes mellitus, gastro-esophageal disease, liver disease, hypertension, asthma, thyroid disease, and ovarian diseases before the interview was administered. Written informed consent was provided by all of the participants in the study, and the design of the study was approved by the Committee on Human Research of Seoul National University Hospital (IRB no., H-0503-144-004).
For continuous variables and categorical variables, t-tests and Pearson's chi-squared tests, respectively, were used to summarize the baseline characteristics of the study population. For associations between each pre-existing disease and the risk of breast cancer, odds ratios (ORs) and 95% confidence intervals (CIs) were calculated by unconditional logistic regression, adjusting for well-known risk factors of breast cancer. Considering frequent comorbid conditions such as diabetes and hypertension, additional analyses were performed by the combination of these two diseases. Further, additional adjustments were done for these two diseases virtually. Age at diagnosis or recruitment, age of menarche, and age of full-term pregnancy were applied in the model as continuous variables, whereas family history of breast cancer and experience of pregnancy were adjusted as categorical variables. Analysis was conducted by stratifying the menopausal status to evaluate the difference in the risk of breast cancer by each pre-existing disease. The test for heterogeneity was conducted for the strata difference. The Cochran's Q was calculated by summing the squared deviations of each estimate from the overall estimate. P for heterogeneity was calculated by comparing the statistic with a χ2 distribution. All statistical analyses were conducted by SAS version 9.3 (SAS Inc., Cary, NC, USA) and STATA version 12 (Stata Corp., College Station, TX, USA).
RESULTS
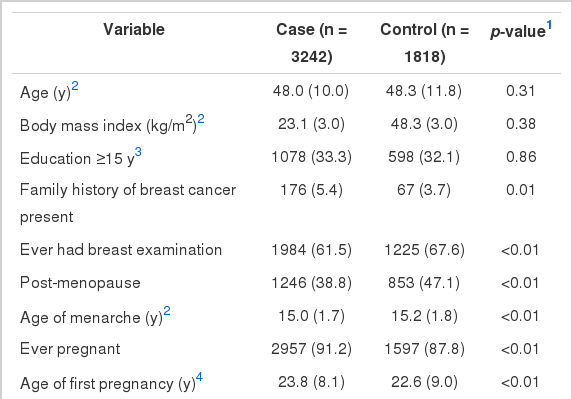
The mean age was 48.0 years for the cases and 48.3 years for the controls. As shown in Table 1, the mean age of menarche was younger in the cases than the controls. Among the cases, there were 91.2% of the participants who ever had pregnancy experience compared to 87.8% of the participants among the controls. Furthermore, 5.4% of the participants among the cases had a family history of breast cancer (3.7% among the controls) and 61.5% of the cases had ever had their breasts examined, including by mammography, ultra-sonography, or manual examination (67.6% among the controls). Additionally, people with breast cancer in this study tended to have smoked cigarettes more than the controls had (7.4% vs. 5.7%).
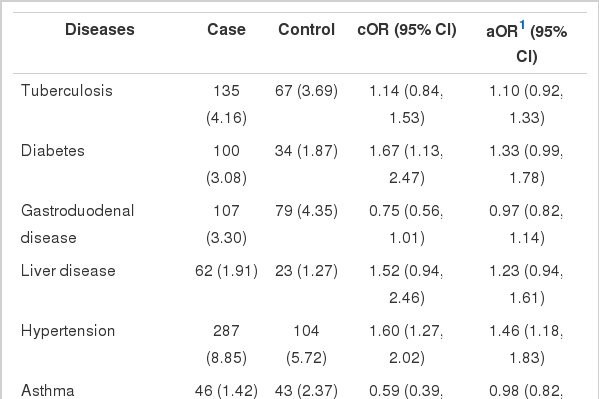
In logistic regression analyses, diabetes (OR, 1.33; 95% CI, 0.99 to 1.78), hypertension (OR, 1.46; 95% CI, 1.18 to 1.83), thyroid diseases (OR, 1.26, 95% CI, 1.00 to 1.58), and ovarian diseases (OR, 1.70; 95% CI, 1.23 to 2.35) were associated with an increased risk of breast cancer when other factors adjusted (Table 2). No other single pre-existing medical condition was associated with breast cancer risk. When people had either diabetes or hypertension or both, it increased the risk of breast cancer significantly (adjusted OR [aOR], 1.54; 95% CI, 1.25 to 1.89, data not shown). However, the risk did not increase significantly in those who both had diabetes and hypertension compared to those who had neither of the conditions (aOR, 1.18; 95% CI, 0.91 to 1.54). For hypertension, the risk was significantly increased after additional adjustment for diabetes (aOR, 1.44; 95% CI, 1.14 to 1.82; data not shown). The risk for diabetes was also higher with additional adjustment for hypertension, but not significantly (aOR, 1.13; 95% CI, 1.14 to 1.82; data not shown).
Table 3 shows the stratified analysis by menopausal status. Although a history of ovarian disease increased the risk of breast cancer with both pre- and post-menopausal status, ovarian disease showed a stronger association in the pre-menopausal population (OR, 4.20; 95% CI, 1.91 to 9.24) than in post-menopausal participants (OR, 1.39; 95% CI, 1.02 to 1.91), and showed significant heterogeneity according to menopausal status (p-heterogeneity 0.01). Pre-existing diabetes increased the risk of breast cancer significantly only in the post-menopause group (OR, 1.44; 95% CI, 1.03 to 2.01), whereas it showed no significant association in the pre-menopause group. Likewise, among the patients who had pre-existing hypertension, only being post-menopausal was significantly associated with an increased risk of breast cancer (OR,1.87; 95% CI, 1.44 to 2.43). For hypertension, the p-heterogeneity was less than 0.01 between the pre- and post-menopausal group. In combination, having either diabetes or hypertension had a stronger association with breast cancer in the post-menopausal group (OR, 1.93; 95% CI, 1.51 to 2.47) than the pre-menopausal group (OR, 1.01; 95% CI, 0.63 to 1.62). However, if the cases were limited to those who had both diabetes and hypertension prior to breast cancer, no significant risk was found in either pre- or post-menopausal groups. In stratified analysis, people who had hypertension or diabetes had a significantly increased risk only in the post-menopausal period (OR, 1.93; 95% CI, 1.51 to 2.47), not in pre-menopause (OR, 1.01; 95% CI, 0.63 to 1.62; p-heterogeneity 0.02). With additional adjustment for hypertension, diabetes was associated with an increased risk in both pre-menopausal (OR, 1.91; 95% CI, 0.64 to 5.71) and post-menopausal (OR, 1.91; 95% CI, 0.64 to 5.71) groups. With hypertension alone, the risk for breast cancer was significantly higher in the post-menopausal group with additional adjustments (OR, 1.84; 95% CI, 1.40 to 2.40), but not the pre-menopausal group (OR, 0.81; 95% CI, 0.48 to 1.34).
DISCUSSION
Our study presents the possibility that certain pre-existing medical conditions are associated with the development of breast cancer, and that these associations differ by a woman's menopausal status.
This study supports previous studies in which a history of hypertension was associated with a decreased breast cancer risk in pre-menopausal women, but the risk was increased in post-menopausal women [20]. Notably, the hypertension alone without comorbid diabetes has been shown to increase the risk of breast cancer significantly. It is suggested that genetic mutations in lymphocytes are more likely to occur in hypertensive women [21]. In an animal study, hypertensive rats were seen to have an abnormal cell cycle [22]. Common risk factors, such as hyperinsulinemia, were also mentioned for both diabetes and hypertension [23]. This could be explained by the effect of insulin, since hypertension is associated with insulin resistance [24]. Even if insulin resistance causes hyperinsulinemia, a different estrogen receptor status according to menopause will have different effects on breast cancer development. As seen in combination analysis with diabetes, the risk increased significantly for those who had ever had hypertension or diabetes. However, in the present study, there was no significantly increased risk of breast cancer in those who both had diabetes and hypertension. Furthermore, in additional adjustment for diabetes, the results were similar to the previous analysis in which diabetes had not been adjusted for. This seems to be the result of the small proportion of participants having these two diseases as comorbid conditions.
In this study, women with diabetes had a significantly increased risk of breast cancer only in the post-menopausal group, but the risk was also increased in the pre-menopausal group, though there was no statistical significance. This might be the result of a lower prevalence of breast cancer in the pre-menopausal group. Although not significant when additionally adjusting for hypertension, there was an increased risk in both the pre-menopause and post-menopause groups. Diabetes seems to increase the risk of breast cancer in both periods. Since they share many risk factors for disease development, the association between diabetes and breast cancer risk has been mentioned in a few previous studies. The results of this study are consistent with many previous findings [25,26]. Many biological factors make the relationship of diabetes and breast cancer development plausible. For example, excess insulin has a mitogenic growth-promoting effect in both normal and malignant breast tissues [27]. In addition, with insulin-like growth factor 1, hyperinsulinemia plays a role in breast cancer development. Insulin decreases sex hormone binding globulin production, which increases free estrogens [28]. Many other mechanisms could possibly explain the relationship between diabetes and breast cancer development. Among them are adipocytokines [29], growth factors [30], and therapies and epigenetic changes from diabetes [31].
Thyroid diseases seem to increase the risk of breast cancer. Our results support previous studies that asserted a relationship between thyroid disorders and breast cancer risk [32,33]. Although this study did not collect more precise information on the specific type of thyroid disease, previous studies have mentioned that goiter and autoimmune thyroiditis are more prevalent in breast cancer patients than the general population in breast cancer patients [34]. There may be interactions between thyroid and breast tissue. They share the trait of concentrating iodine, and both tissues have abundant thyroid-stimulating hormone receptors [35]. Since no information on specific medication of thyroid diseases was collected, it was not possible to evaluate the role of 131I in breast carcinogenesis, which other studies have not determined conclusively.
Among ovarian diseases without malignancy, ovarian cysts are common. A few studies have reported that ovarian cyst formation is associated with high serum estradiol levels and that menstruation also affects cyst development [36]. It seems that a high estrogen level with continued menstruation is related to ovarian cyst formation, and this is also related to breast cancer development [37]. Another relatively common disease of the ovary is endometriosis, which is also estrogen-dependent [38]. Endometriosis is said to have multiple etiologies, including genetic, hormonal, and immunological factors [39]. Endometriosis causes infertility, which leads to nulliparity or delayed childbearing [38], which are also risk factors for breast cancer [6]. Furthermore, treatment of endometriosis with progestin, danazol, or oral contraceptives could trigger the development of breast cancer [40]. Both endometriosis and breast cancer have a hormone-dependent etiology.
As information was collected only by questionnaire, misclassification of previous disease history could have occurred. In particular, as some of the items on previous medical history were asked in non-specific disease terms, such as 'gastroduodenal disease,' 'thyroid disease,' or 'ovarian disease,' the participants might not have been certain of their disease status.. Additionally, by using broad terms for each pre-existing condition, it was not possible to analyze the disease categories in more detail. Confirming diseases by checking medical records would have strengthened the study. The relatively small number of cases of each pre-existing disease when comparing the pre-menopausal and post-menopausal groups was also a potential limitation. Since this may have caused a reduction in the statistical power, especially in the pre-menopausal group, results with non-statistical significance should also be taken into consideration. Nevertheless, data on the association of hypertension and ovarian diseases with breast cancer development should be considered significant, considering the magnitude of the OR.
In conclusion, our study has identified medical conditions that have a significant association with breast cancer development, which can differ by the menopausal status of women. Hypertension, ovarian disease, diabetes, and thyroid disease significantly increased the risk of breast cancer. In a stratified analysis by menopausal status, hypertension showed statistical significance of increased development of breast cancer only in the post-menopausal population. Ovarian disease showed an increased risk of breast cancer, and the OR was elevated more in the pre-menopause group. The findings in this study implicate certain medical conditions in the development of breast cancer. Further study is needed to evaluate their biological mechanisms and relationships with other diseases.
ACKNOWLEDGMENTS
This research was supported by the Basic Research Laboratory (BRL) program through the National Research Foundation of Korea funded by the Ministry of Education, Science and Technology (2012-0000347).
Notes
The authors have no conflicts of interest with the material presented in this paper.